Ions In Solution And Their Solvation: Unraveling the Mysteries of Aqueous Electrolytes

Understanding Ions in Solution
4.6 out of 5
| Language | : | English |
| Paperback | : | 32 pages |
| Item Weight | : | 1.73 ounces |
| Dimensions | : | 5.5 x 0.08 x 8.5 inches |
| File size | : | 11962 KB |
| Text-to-Speech | : | Enabled |
| Screen Reader | : | Supported |
| Enhanced typesetting | : | Enabled |
| Print length | : | 302 pages |
| Lending | : | Enabled |
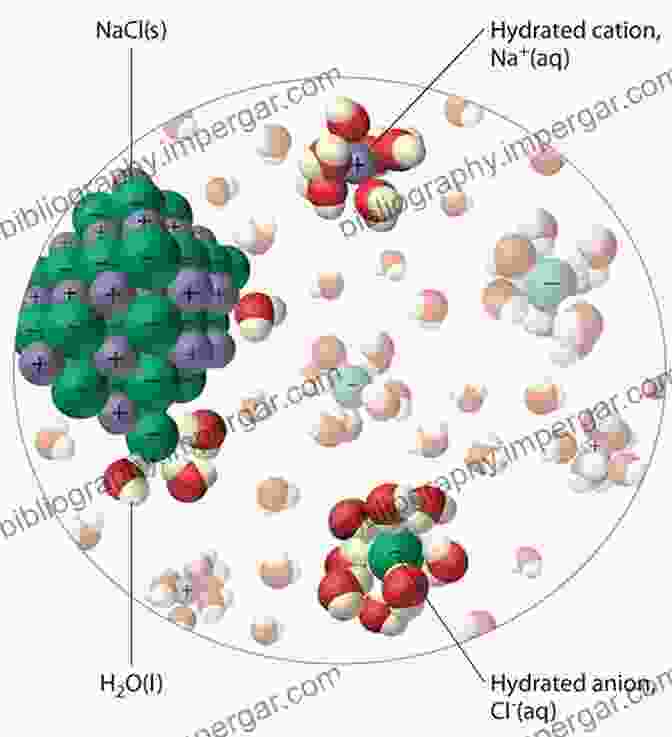
Ions, the charged particles that form when atoms or molecules gain or lose electrons, play a crucial role in various chemical and biological processes. When these ions are dissolved in a solvent, they create a solution known as an electrolyte. In particular, aqueous electrolytes, where water serves as the solvent, are ubiquitous in nature and have immense practical significance.
The behavior of ions in solution is governed by their interactions with the surrounding solvent molecules. These interactions, termed solvation, profoundly influence the properties and reactivity of the ions. Understanding the mechanisms and consequences of solvation is essential for comprehending the behavior of electrolytes and their applications.
Solvation Mechanisms: Unveiling the Hidden Forces
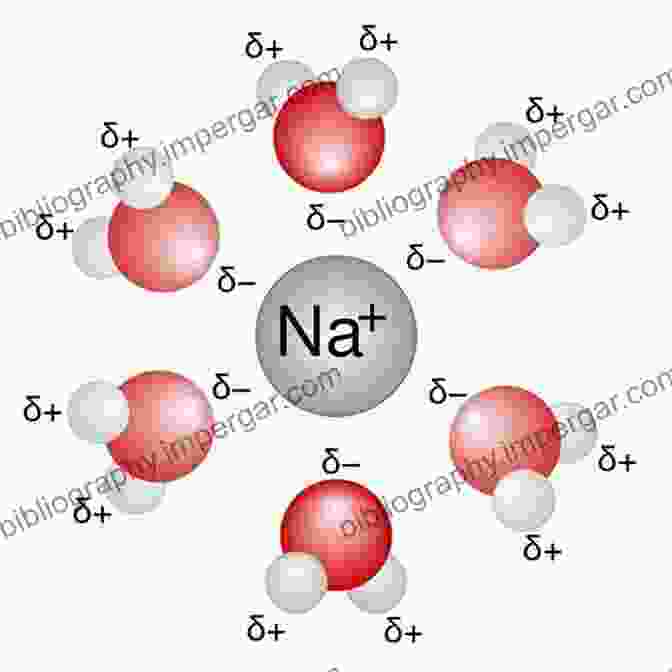
Solvation involves the formation of a solvation shell around the ion, consisting of solvent molecules that are electrostatically attracted to the ion's charge. This shell of solvent molecules shields the ion from direct contact with other ions or solvent molecules, thereby affecting its reactivity and transport properties.
Two primary solvation mechanisms exist: electrostatic solvation and specific solvation. Electrostatic solvation occurs due to the direct electrostatic attraction between the ion and the solvent molecules' permanent dipoles or induced dipoles. Specific solvation, on the other hand, involves the formation of specific interactions, such as hydrogen bonding or ion-dipole interactions, between the ion and the solvent molecules.
Consequences of Solvation: Shaping Ion Behavior
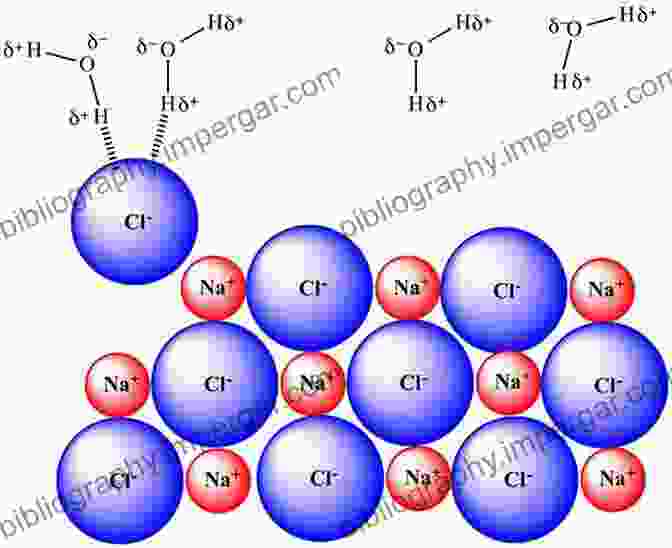
Solvation has profound consequences for the behavior of ions in solution. It can alter the ion's effective size, influencing its transport properties and interactions with other ions or molecules. Additionally, solvation affects the ion's reactivity, influencing its ability to participate in chemical reactions.
The extent and nature of solvation depend on various factors, including the charge and size of the ion, the polarity and hydrogen-bonding ability of the solvent, and the temperature. Understanding these factors is crucial for predicting and controlling the behavior of ions in solution.
Applications of Ions in Solution: From Batteries to Medicine
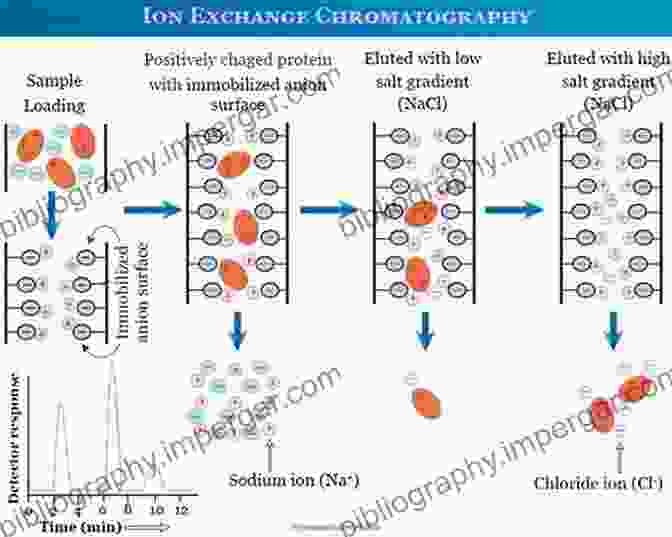
Ions in solution have a wide range of applications in various fields, including electrochemistry, materials science, and biology. Electrolyte solutions are essential components of batteries, enabling the storage and release of electrical energy. They are also used in electrolysis, a process that utilizes electrical energy to drive chemical reactions, and electroplating, a technique used to coat surfaces with a thin layer of metal.
In biology, ions play a critical role in maintaining osmotic balance, regulating nerve impulses, and facilitating muscle contractions. Understanding the behavior of ions in biological systems is essential for comprehending fundamental physiological processes and developing therapeutic interventions.
: Ions in Solution: A Cornerstone of Scientific Discovery
The exploration of ions in solution has been a cornerstone of scientific discovery, leading to groundbreaking insights into chemical and biological processes. By unraveling the intricate interactions between ions and solvents, scientists have developed a comprehensive understanding of the behavior of electrolytes and their applications in various fields.
The ongoing research in this domain promises to further expand our knowledge and lead to even more transformative discoveries. With the advancement of experimental techniques and computational modeling, we can expect to gain deeper insights into the solvation processes, ion transport mechanisms, and the role of ions in complex biological systems.
4.6 out of 5
| Language | : | English |
| Paperback | : | 32 pages |
| Item Weight | : | 1.73 ounces |
| Dimensions | : | 5.5 x 0.08 x 8.5 inches |
| File size | : | 11962 KB |
| Text-to-Speech | : | Enabled |
| Screen Reader | : | Supported |
| Enhanced typesetting | : | Enabled |
| Print length | : | 302 pages |
| Lending | : | Enabled |
Do you want to contribute by writing guest posts on this blog?
Please contact us and send us a resume of previous articles that you have written.
 Book
Book Novel
Novel Page
Page Chapter
Chapter Text
Text Story
Story Genre
Genre Reader
Reader Library
Library Paperback
Paperback E-book
E-book Magazine
Magazine Newspaper
Newspaper Paragraph
Paragraph Sentence
Sentence Bookmark
Bookmark Shelf
Shelf Glossary
Glossary Bibliography
Bibliography Foreword
Foreword Preface
Preface Synopsis
Synopsis Annotation
Annotation Footnote
Footnote Manuscript
Manuscript Scroll
Scroll Codex
Codex Tome
Tome Bestseller
Bestseller Classics
Classics Library card
Library card Narrative
Narrative Biography
Biography Autobiography
Autobiography Memoir
Memoir Reference
Reference Encyclopedia
Encyclopedia Jan Timman
Jan Timman Esther Safran Foer
Esther Safran Foer Kevin Nugent
Kevin Nugent Forbes W Robertson
Forbes W Robertson Frederick Taylor
Frederick Taylor Frederick N Martin
Frederick N Martin Frank J Andruss Sr
Frank J Andruss Sr Mark Stephen Clifton
Mark Stephen Clifton Francis Bacon
Francis Bacon Garry Boulard
Garry Boulard James W Fatherree
James W Fatherree Alisdair Aird
Alisdair Aird Sherianna Boyle
Sherianna Boyle Robert W Hastings
Robert W Hastings Frank Warren Coburn
Frank Warren Coburn Fab Chris
Fab Chris Frances Howard Snyder
Frances Howard Snyder Frank H P Fitzek
Frank H P Fitzek Hermann Bengtson
Hermann Bengtson Gabriel Marcel
Gabriel Marcel
Light bulbAdvertise smarter! Our strategic ad space ensures maximum exposure. Reserve your spot today!

 Robert HeinleinUnleash the Power of Extraordinary Minds: "The Brains That Enabled Victory"
Robert HeinleinUnleash the Power of Extraordinary Minds: "The Brains That Enabled Victory" Edmund HayesFollow ·3.2k
Edmund HayesFollow ·3.2k Emilio CoxFollow ·5.4k
Emilio CoxFollow ·5.4k Oscar BellFollow ·19.1k
Oscar BellFollow ·19.1k Charles BukowskiFollow ·5.1k
Charles BukowskiFollow ·5.1k Oliver FosterFollow ·9.9k
Oliver FosterFollow ·9.9k Bernard PowellFollow ·16.5k
Bernard PowellFollow ·16.5k Stuart BlairFollow ·19.3k
Stuart BlairFollow ·19.3k Jayden CoxFollow ·7.8k
Jayden CoxFollow ·7.8k

 Alexander Blair
Alexander BlairBecoming Sports Agent Masters At Work: The Ultimate Guide
What is a Sports...

 Xavier Bell
Xavier BellUnveiling the Enchanting World of Upper Bohemia: A Review...
A Captivating...

 Chris Coleman
Chris ColemanUnveiling the Secrets: Extreme Rapid Weight Loss Hypnosis...
In the relentless pursuit of a slimmer,...
4.6 out of 5
| Language | : | English |
| Paperback | : | 32 pages |
| Item Weight | : | 1.73 ounces |
| Dimensions | : | 5.5 x 0.08 x 8.5 inches |
| File size | : | 11962 KB |
| Text-to-Speech | : | Enabled |
| Screen Reader | : | Supported |
| Enhanced typesetting | : | Enabled |
| Print length | : | 302 pages |
| Lending | : | Enabled |















